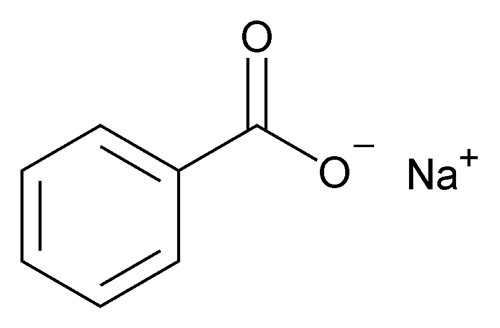
Structure
-Chemical formula of NaC7H5CO2
-Consists of:
-Consists of:
- A benzene ring, with hydrogen around 6 of the carbons
- Another Carbon bonded the the benzene ring, and double bonded to an oxygen, as well as a single bond to another oxygen ion
- An ionic bond between a sodium ion and the oxygen ion.
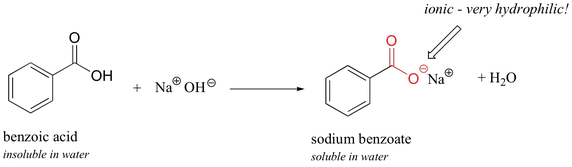
How it is created
Benzoic acid reacting with sodium hydroxide create sodium benzoate along with water
properties
-White flakes/powder,
-Ionic bond between the sodium ion and oxygen ion makes the coupound more soluable in water than Benzoic Acid
-Lowers the ph of a substance when mixed to a ph of 2.5-4
-Combined with ascorbic acid may created traces of benzene, a carcinogen
-Highly flammable and easily conbustible
Sci Show on Sodium Benzoate with Citric acid (7:17-8:32)
-Ionic bond between the sodium ion and oxygen ion makes the coupound more soluable in water than Benzoic Acid
-Lowers the ph of a substance when mixed to a ph of 2.5-4
-Combined with ascorbic acid may created traces of benzene, a carcinogen
-Highly flammable and easily conbustible
Sci Show on Sodium Benzoate with Citric acid (7:17-8:32)